pH 측정기 소개
pH 측정기의 정의
pH 측정기는 용액의 pH 값을 결정하는 데 사용되는 도구를 말합니다. pH 측정기는 갈바니 배터리의 원리로 작동합니다. 갈바니 전지의 두 전극 사이의 기전력은 Nerns의 법칙을 기반으로 하며 이는 전극의 특성뿐만 아니라 용액 내 수소 이온 농도와도 관련이 있습니다. 1차 전지의 기전력과 수소이온 농도 사이에는 대응 관계가 있으며, 수소이온 농도의 음의 로그값이 pH 값이 됩니다. pH 측정기는 농업, 환경 보호 및 산업 분야에서 널리 사용되는 일반적인 분석 기기입니다. 토양 pH는 토양의 중요한 기본 특성 중 하나입니다. pH 측정 중에는 테스트할 용액의 온도 및 이온 강도와 같은 요소를 고려해야 합니다.
pH 측정기의 원리
pH는 수용액의 수소 이온 농도의 음의 로그로 정의됩니다. 복잡하게 들리지만 매우 간단한 용어로 pH는 용액의 산도 또는 알칼리도를 정량화하는 데 사용되는 숫자입니다. 숫자는 특정 물질이 용액에서 방출할 수 있는 수소 이온의 수를 나타냅니다. pH 범위에서 pH 7은 중성으로 간주됩니다. pH가 0~7인 용액은 산성으로 간주되고, 7~14 이상의 용액은 알칼리성 용액이라고 합니다. 생물학적 시스템에서 pH는 매우 중요합니다. 세심하게 조절된 pH 덕분에 우리 몸에 있는 대부분의 생체분자는 탁월한 기능을 수행할 수 있습니다. 실험 시스템에서도 정확한 결과를 얻으려면 필요한 pH를 유지해야 합니다. 따라서 생물학적 실험에서는 pH 측정기라는 장치를 사용하여 pH를 주의 깊게 모니터링합니다.
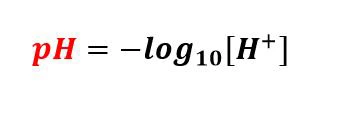
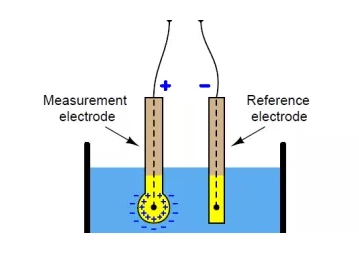
pH 측정기는 용액 내 수소 이온의 활성을 측정하고 이 정보를 전송하는 pH 반응 전극입니다. 이 장치는 두 개의 유리관으로 구성되어 있으며 각 관에는 전극, 기준 전극 및 센서 전극이 포함되어 있습니다. 기준 전극은 포화 KCl 용액으로 만들어졌고, 센서 전극은 pH 7의 완충 용액을 포함하고 염화은으로 코팅된 은선을 이 두 용액에 담근다. 센서 전극 끝에는 실리카와 금속염으로 코팅된 다공성 유리로 만들어진 전구가 있습니다.
용액의 pH를 측정하기 위해 pH 측정기를 용액에 담급니다. 센서 전극의 전구가 용액에 접촉하면 용액의 수소 이온이 전구의 금속 이온을 대체합니다. 금속 이온의 이러한 치환으로 인해 금속 와이어에 전류가 흐르고 전압계로 판독됩니다.
pH 측정기는 생물학 실험실에서 가장 널리 사용되는 장비 중 하나입니다. 완충액, 용액 및 시약의 pH를 정기적으로 분석하여 실험 조건이 올바른지 확인합니다. 정확한 판독값을 보장하려면 장비를 정기적으로 교정해야 합니다.
PH 측정기 감지기 적용
국내 하수처리 공정에 PH 측정기 검출기 적용

전기 도금 폐수 처리에 pH 측정기 적용

산업에서의 온라인 PH 측정기 적용

PH 측정기 교정
